 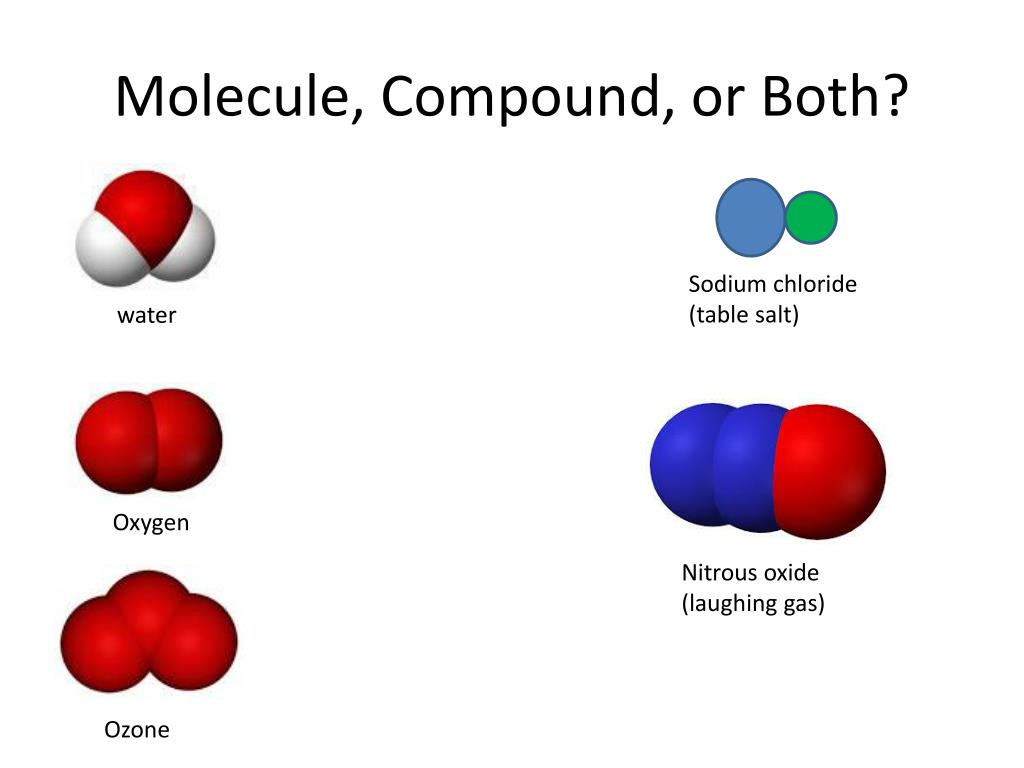
It reacts with all metals except gold and platinum. Pure sulfur is a tasteless, odourless, brittle solid that is pale yellow in colour, a poor conductor of electricity, and insoluble in water. Example: the empirical formula for the compound is C 3H 8, then its molecular formula might be C 3H 8, C 6H 16, etc. sulfur (S), nonmetallic chemical element belonging to the oxygen group (Group 16 VIa of the periodic table), one of the most reactive of the elements. It is based on the exact number of atoms for each type present in the compound. On the other hand, the molecular formula can be counted as the multiple of the empirical formula. Elaborate the difference between molecular formula and empirical formula with Example.Īns: The empirical formula can be termed as the simplest formula for any compound. Further, Salt (NaCl) resembles an ionic compound because it is made from more than one type of element which is held together by ionic bonds. Water (H 2O) is a molecular compound because it is a substance made from more than one kind of element that is held together by molecular bonds. N 2 can be seen as a molecule since the bond formed between the nitrogen atoms depicts the molecular bond. Keep a note that, molecules contain molecular bonds. To be clear, electrons are permitted to be shared between atoms or electrons can be totally removed from a single atom and transferred to another. For precise knowledge, something which is a molecule doesn’t depend on the type of bond created when atoms join together. This is because they only contain a single type of element. On the other hand, nitrogen gas (N 2), as well as buckminsterfullerene (C 60), cannot be classified as compounds. These are called as compounds since each of them contain more than one type of element. Table salt (NaCl), Water (H 2O), carbon dioxide (CO 2), chlorophyll (C 55H 72O 5N 4Mg) etc., are several examples of compounds. To be precise, a compound is basically a substance which is composed using two or more different elements. Difference Between a Compound and Molecule Some more examples of compounds include table salt (NaCl), chalk (CaCO 3) and water (H 2O). Similarly, the chemical formula for carbon dioxide is CO 2, and it is formed using two elements, namely carbon and oxygen. Hence, those substances made by the merger of two or more types of elements are Compounds.Ītoms present in a compound are chemically connected and hence cannot be separated easily. Two different elements combine, giving birth to a new substance called as water. In this note, water is made from 2 hydrogen atoms and 1 oxygen atom. Remember that, the chemical formula for water is H 2O. Do we consider it as a molecule or compound? Whenever two or more atoms of the same element merge together, we tag them as molecules. Since molecules are built out of atoms which are jointly held via chemical bonds, there are chances of significant variance in terms of size and complexity.įor example, oxygen holds a molecular formula O 2. It delivers the same chemical properties of that particular compound. The type of bonds holding elements together in a compound can vary: two common types are covalent bonds and ionic bonds. Water: two hydrogen atoms bonded to an oxygen atom. In mixtures, the substances present are not chemically bonded together. A molecule can be termed as the smallest unit associated with a chemical compound. A compound is a substance formed when two or more chemical elements are chemically bonded together. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
